Tech Deep Dive: Graphite vs Silicon Anodes – Materials, Supply Chains, and Risk
Electric vehicle and stationary storage programs are no longer debating whether to use lithium-ion batteries; the debate has moved inside the cell. Anode chemistry, in particular the balance between graphite and silicon, has become a structural determinant of range, fast-charge capability, and-critically-supply chain risk. This is the terrain of the current “Tech Deep Dive: Graphite vs Silicon Anodes – Materials, Supply Chains, and Risk.”
Graphite has delivered the reliability that made lithium-ion bankable at scale. Silicon, with roughly an order-of-magnitude higher theoretical capacity, is now the front-runner for the next energy-density step-change. Yet the trade-off is clear: graphite is technologically mature but geopolitically exposed; silicon is geographically diversified but technically immature at volume.
Materials Dispatch views this not as a simple technology upgrade, but as a restructuring of risk: from a single dominant material with concentrated refining to a mixed anode landscape where chemistry choice, supplier footprint, and policy alignment interact in non-trivial ways.
1. Material Fundamentals: What Graphite and Silicon Actually Do in the Cell
1.1 Graphite: The Workhorse Intercalation Host
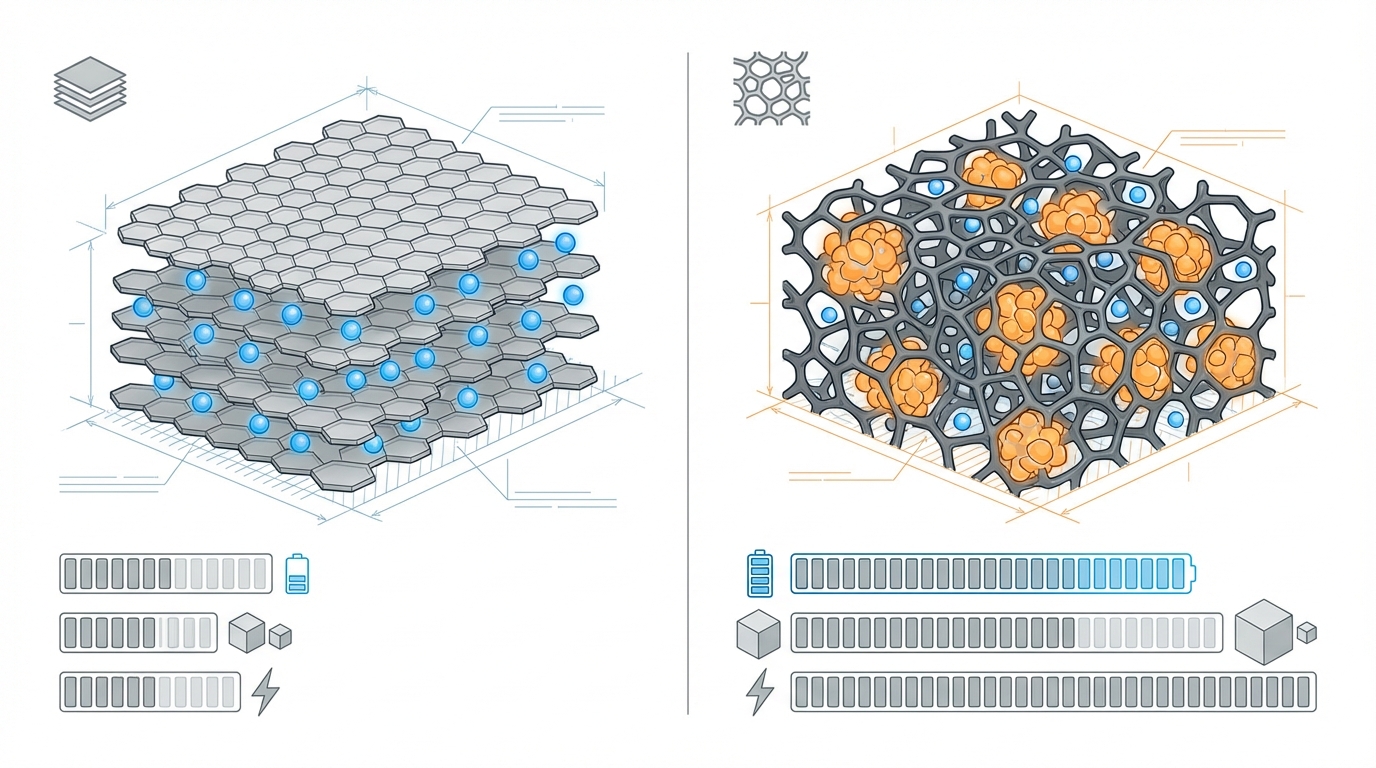
Graphite anodes rely on lithium intercalation. Lithium ions insert between the graphene layers to form stages of LixC6, with a commonly cited theoretical capacity of around 372 mAh/g for fully lithiated graphite. In practice, commercial anodes operate somewhat below this limit to preserve cycle life and avoid lithium plating.
The key operational strength of graphite is dimensional stability. During cycling, well-designed graphite anodes typically experience limited volume change, enabling thousands of cycles in mainstream EV duty cycles when paired with appropriate cathodes and operating windows. This stability simplifies mechanical design of cells and packs, reduces mechanical stress on separators, and limits continuous re-formation of the solid-electrolyte interphase (SEI).
The downside is energy density and fast-charge headroom. Graphite’s specific capacity caps the anode-side contribution to cell-level energy density. Under aggressive fast-charging, the anode potential can reach levels where lithium plating becomes a dominant failure mode, degrading cycle life and raising safety concerns. Most current-generation fast-charge EV strategies so rely on sophisticated thermal management and charge protocols to protect graphite rather than fundamentally changing the anode material.
1.2 Silicon: From Intercalation to Alloying
Silicon operates through a different mechanism: lithium-silicon alloying rather than intercalation. Fully lithiated silicon (approaching Li15Si4) has a widely cited theoretical capacity on the order of 3,500-3,600 mAh/g, almost an order of magnitude higher than graphite. Even partial utilization of this capacity enables significant anode mass reduction and higher cell-level energy density.
However, alloying drives extreme volume change-often cited on the order of several hundred percent between fully lithiated and delithiated states. This expansion-contraction cycle induces mechanical stress, cracking of silicon particles, loss of electrical contact, and continuous SEI growth as fresh surface is exposed. The result, in unmitigated form, is rapid capacity fade and gas evolution.
Modern silicon-anode approaches rarely rely on bulk silicon particles. Instead, they use engineered structures—nanowires, nano- or micro-structured silicon, silicon-oxide (SiOx), or silicon embedded in carbon frameworks—combined with optimized binders and electrolyte additives. Commercial players such as Amprius, Group14, Sila Nanotechnologies, NanoGraf, and others each pursue distinct architectures, but they all converge on the same challenge: harnessing silicon’s capacity while controlling mechanical and interfacial damage.
1.3 Comparing Performance Levers: Energy, Power, and Life
At cell level, the graphite–silicon trade-off is not binary. Most near-term implementations use hybrid anodes with a fraction of silicon blended into graphite, typically in the single-digit to low double-digit percentage range by weight. This approach targets incremental energy-density improvements while retaining established manufacturing baselines and cycle life expectations.
Various public demonstrations by silicon-anode developers have reported energy densities significantly above those of standard graphite-based cells, with some lab and early commercial cells claiming gravimetric energy densities around or beyond 400 Wh/kg in specialized formats. Traditional EV-grade graphite-based cells often cluster materially below that figure, depending on cathode chemistry and format. The exact numbers vary by chemistry, packaging, and operating window, but the direction of travel is consistent: silicon increases the ceiling.
Fast charging is another axis. Silicon-containing anodes can, in principle, accept higher currents because the anode potential can be maintained at safer levels while storing more lithium. Several developers publicly highlight sub-15-minute charge profiles under specific conditions. Yet high-power protocols also accelerate mechanical and SEI-related stress in silicon, so fast-charge capability is coupled tightly to thermal management, cell design, and control strategies rather than anode material alone.
1.4 Property Comparison in Operational Terms
| Parameter | Graphite-Dominant Anode | Silicon-Enhanced / Silicon-Dominant Anode | Operational Consequence |
|---|---|---|---|
| Theoretical specific capacity (mAh/g) | ~372 (lithiated graphite) | ~3,000–3,600 (lithiated silicon, depending on phase) | Silicon enables substantially higher anode-side capacity in principle. |
| Volume change during cycling | Typically limited; often quoted at <10% range | Very high; often cited in the several-hundred-percent range for pure Si | Silicon requires advanced mechanical accommodation to avoid fracture. |
| Cycle life (EV-grade duty) | Established; multi-thousand cycles feasible with optimized cells | Highly architecture-dependent; commercial targets focus on matching graphite-grade warranties | Long-term stability of silicon solutions remains a core qualification question. |
| Fast-charge tolerance | Constrained by lithium plating risk at high C-rates | Potentially higher acceptance if mechanical/SEI issues managed | Silicon blends are being positioned as fast-charge enablers, but validation is ongoing. |
| Manufacturing maturity | Highly mature; global multi-GWh scale across regions | Emerging; pilot to early GWh-scale lines focused in North America, Europe, and East Asia | Graphite remains baseline; silicon capacity ramps from a lower base. |
The pattern is clear: silicon pushes the frontiers on energy density and potentially charge rate, while graphite anchors mechanical stability and proven lifetime. The technology race is not only about which material is “better,” but about which combinations deliver acceptable performance at industrial scale with manageable risk.
2. Graphite Supply Chains: From Geology to Export Controls
2.1 Upstream: Natural vs Synthetic Graphite
Graphite for lithium-ion anodes comes from two broad sources: natural graphite mined from deposits, and synthetic graphite produced from petroleum coke or other carbonaceous precursors at high temperatures. Natural graphite offers lower energy intensity in mining but requires extensive processing and purification. Synthetic graphite offers highly controlled properties but is energy- and emission-intensive due to graphitization furnaces operating at very high temperatures.
From an operational standpoint, natural graphite anode feedstock is typically produced by crushing and flotation to concentrate carbon, followed by micronization, spheronization, and purification to reach battery-grade specifications. This route relies on water-intensive beneficiation and often uses chemical or thermal purification steps to reduce impurities to low ppm ranges. Synthetic graphite, in contrast, begins from petroleum or coal-derived coke that is calcined, formed into shapes, and then graphitized. The process consumes significant electricity and can have a sizable CO2 footprint, depending on power sources.
For EV-cell producers, both routes converge midstream: the critical step is spherical graphite production with controlled particle size distribution, tap density, surface area, and coating, rather than whether the carbon originated from rock or coke. However, the geographic and regulatory profile of these routes differs sharply, and that distinction is increasingly important.
2.2 Midstream: Spherical Graphite and Chinese Dominance
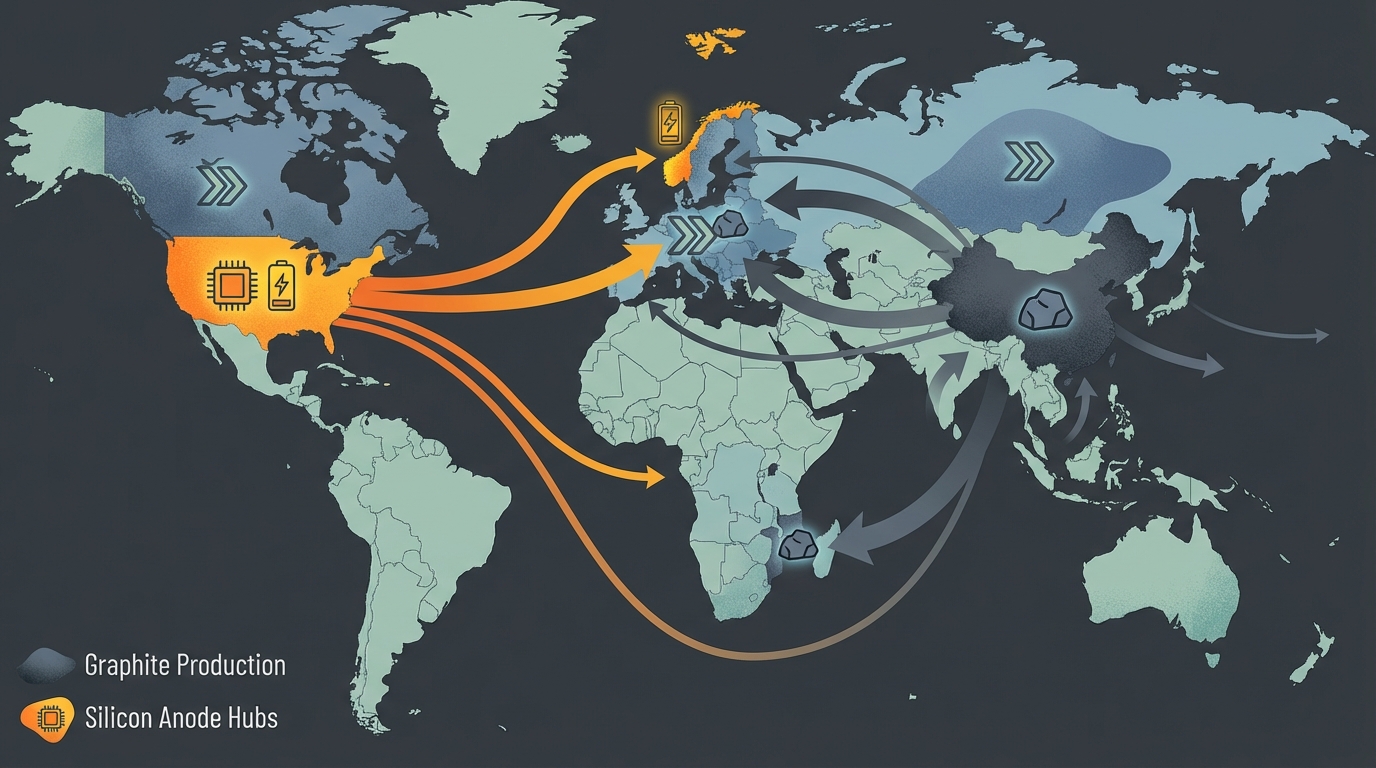
Most of the world’s anode-grade spherical graphite refining capacity is currently located in China. Public data from agencies such as USGS and industry trackers consistently indicate that China accounts for a majority of natural graphite mining and an even larger share of battery-grade graphite processing, often cited at well over four-fifths of global output. Even when ore is mined elsewhere—Mozambique, Canada, Madagascar, or other jurisdictions—a substantial fraction has historically been shipped to China for purification and spheronization.
The refining process is where much of the value-add and environmental impact arises. Chemical purification routes frequently use hydrofluoric acid and other reagents to lower impurity levels, raising worker safety and effluent management challenges. Thermal purification requires high-temperature furnaces and significant electricity. Many Chinese facilities have spent years optimizing throughput, yields, and cost structures across these steps, building a high-barrier-to-entry competitive moat.
This concentration creates a simple but uncomfortable reality for battery producers in North America, Europe, and allied jurisdictions: even with diversified mining, the system remains exposed to Chinese refining policy, permitting cycles, and local environmental enforcement. Export licensing regimes and critical minerals lists have turned what used to be a procurement detail into a board-level risk item.
2.3 Downstream: Anode Production and Qualification Cycles
Downstream of spherical graphite and related precursors, anode manufacturers mix active materials with binders, conductive additives, and solvents, coat copper foil, dry and calendar the electrodes, and then cut, stack, or wind them into cells. This is also where new non-Chinese capacity is scaling: several facilities in North America and Europe are seeking to process imported natural graphite or domestically produced synthetic graphite into finished anodes without intermediate Chinese steps.
However, anode production is tightly integrated into cell manufacturers’ qualification regimes. Changing supplier, surface coating, or particle morphology can require extensive requalification, including formation cycle optimization and long-term durability tests. This slows diversification. Even when alternative graphite sources are available, ramping them into qualified EV cells is a multi-year operational exercise, not a simple sourcing switch.
2.4 Regulatory and ESG Pressures on Graphite
Environmental regulation is tightening around both natural and synthetic graphite. For natural graphite, water use, tailings management, and biodiversity impact at mine sites are focal points for permitting agencies and local communities. For refining, chemical usage and disposal are under increasing scrutiny. For synthetic graphite, greenhouse-gas intensity of production is a growing concern for automotive OEMs that have lifecycle emissions targets.
Policy instruments such as the US Inflation Reduction Act’s “foreign entity of concern” provisions and the EU’s Critical Raw Materials Regulation are not merely labels; they influence where anode-grade graphite can be counted towards local-content thresholds and subsidies. In practice, this adds another layer of complexity: some graphite volumes are technically available on the market but effectively constrained for certain EV programs due to origin and processing history.
In short, graphite is abundant as an element and technologically mature as an anode, yet its refined form is now entangled in policy, ESG, and industrial strategy debates. The risk is less about physical scarcity and more about concentration, compliance, and the pace at which new refining capacity outside China can reach competitive cost and quality.
3. Silicon Anode Supply Chains: Abundant Element, Scarce Processing
3.1 Raw Materials: From Quartz to Metallurgical Silicon
Silicon, the second most abundant element in the Earth’s crust, is not constrained at the ore level. Quartz and other silica-rich materials are globally distributed, and metallurgical-grade silicon (MG-Si) is produced at large scale in multiple regions, including North America, Europe, and Asia. MG-Si is already used extensively in aluminum alloys, chemicals, and solar-grade silicon production.
For anodes, however, the bottleneck is not MG-Si itself but the conversion of silicon into nano- or microstructured forms with tightly controlled properties. Battery-grade silicon materials require narrow particle sizes, engineered porosity, specific surface chemistries, and controlled impurity profiles. These are produced through processes such as gas-phase deposition (for nanowires), high-energy milling, plasma synthesis, or various proprietary methods.
As a result, the upstream silicon resource base offers comfort from a physical availability perspective, but the midstream processing layer is nascent and capital intensive. The leverage point in the silicon anode supply chain is not the mine but the specialized processing plant.
3.2 Midstream Technologies: Multiple Pathways, Common Constraints
Silicon-anode midstream players are pursuing diverse technical paths:
- Silicon nanowires: Grown via chemical vapor deposition on conductive scaffolds, this approach (seen in public descriptions from companies like Amprius) aims to accommodate volume expansion along the wire axis while maintaining electrical contact.
- Silicon-carbon composites: Silicon particles embedded in porous or graphitic carbon matrices (e.g., architectures promoted by Group14 and others) distribute stresses and buffer expansion while leveraging carbon’s conductivity.
- Silicon-oxide and silicon-rich oxides: SiOx-based materials offer lower effective expansion and more gradual lithiation profiles at the cost of reduced specific capacity vs pure silicon.
- Graphite–silicon hybrids: Blending silicon into graphite with tailored binders and coatings yields incremental performance gains with lower disruption to existing anode lines.
Each route imposes different capex and opex structures. Gas-phase processes using silane or other precursors raise stringent safety, gas-handling, and permitting requirements. Solid-state routes can be more modular but still demand advanced powder-handling, classification, and surface-treatment equipment. Across all approaches, tight process control is essential: small deviations in particle morphology or surface chemistry can translate into large swings in cycle life and gas generation.
3.3 Geographic Footprint: A More Distributed Base
Unlike graphite refining, silicon-anode midstream capacity is more geographically distributed from inception. Many of the leading developers are headquartered or building major facilities in the United States and Europe, often co-located with existing semiconductor, specialty-chemicals, or advanced-materials clusters. East Asia remains critical, particularly for integration with established cell manufacturing ecosystems in Korea, Japan, and China, but the vendor base is not as singularly concentrated as graphite refining.
Moreover, government support programs in North America and Europe explicitly target silicon-anode capacity as part of broader battery-industrial strategies. Grants, loans, and tax incentives are being deployed to bridge initial capex gaps, recognizing that silicon processing plants resemble specialty chemical or semiconductor facilities in their complexity and safety requirements.
This positioning creates an interesting asymmetry: silicon anodes are technically more challenging yet politically favored, while graphite is technically mature yet increasingly scrutinized. The net result is a supply landscape where growth in silicon capacity is likely to be policy-pulled and regionally diversified, even if starting from a much smaller base than graphite.
3.4 Technology Maturity and Qualification Risk
For OEMs and cell manufacturers, the main constraint on silicon is not raw material availability but technology maturity and qualification risk. Public announcements from silicon-anode companies often showcase high energy densities and promising cycle life in cell formats targeted at aerospace, premium EVs, or consumer electronics. Translating those results into multi-GWh automotive lines involves several non-trivial steps:
- Scaling from pilot to production while preserving particle morphology and surface chemistry.
- Ensuring binder and electrolyte systems remain stable under manufacturing variability.
- Meeting stringent safety and abuse-test standards, including nail penetration, thermal runaway characterization, and crush tests.
- Proving calendar life under real-world temperature and state-of-charge distributions, not just cycling at laboratory conditions.
Early silicon deployments in EVs are appearing first in higher-end or specialty models, where the performance premium justifies higher material costs and where volumes are moderate enough to manage supply risk. Over time, silicon-graphite blends are expected to diffuse into more mainstream platforms if durability and cost trajectories align with OEM requirements.
4. Comparative Risk Map: Material, Geopolitics, Technology, ESG
4.1 Material Availability and Concentration Risk
From a pure resource perspective, both carbon and silicon are abundantly available in the Earth’s crust. The relevant risk is not geological scarcity but the industrial structure of conversion to battery-grade materials.
Graphite’s risk locus is refining concentration. A large share of the world’s spherical graphite capacity sits in a single country, which has already demonstrated willingness to apply export controls and industrial policy in other critical-materials sectors. Any disruption—whether from policy, environmental enforcement, or local energy constraints—can ripple through EV and storage programs globally.
Silicon’s risk locus is processing maturity. The abundance of silica and global MG-Si production offers comfort on upstream availability, but the specialized plants turning silicon into advanced anode materials are few, young, and technology-specific. If a key vendor’s process underperforms in the field, or if scaling reveals unforeseen reliability issues, substitution options are more limited in the short term.
4.2 Geopolitical and Trade Exposure
Graphite’s heavy processing concentration in China exposes it directly to the evolving landscape of export controls, tariffs, and local-content rules. Measures that tighten export licensing or reclassify certain graphite products as sensitive can affect availability and qualification timelines far beyond what raw tonnage statistics might suggest.
Silicon-anode supply chains, anchored more strongly in North America and Europe alongside East Asian partners, align more naturally with Western industrial-policy objectives. This does not eliminate geopolitical risk—technology export controls and cross-border investment review can also affect silicon technologies—but the risk is more distributed across jurisdictions rather than concentrated in one.
One structural insight emerges here: the geopolitical risk of graphite is about single-node concentration; the geopolitical risk of silicon is about many small nodes whose reliability is not yet fully proven. In other words, graphite’s threat is macro and concentrated, silicon’s is micro and distributed.
4.3 Technology and Scaling Risk
Graphite technology is well understood. Known issues—such as lithium plating at high charge rates or gas evolution under certain conditions—are familiar and embedded in existing design rules. Scaling risk for new graphite capacity primarily relates to building and commissioning purification and shaping lines, not to fundamental uncertainty about anode behavior.
Silicon technology, in contrast, still sits on a steeper learning curve. Each vendor’s architecture (nanowire vs composite vs SiOx, and others) has distinct failure modes and sensitivities. Binder chemistries, electrolyte formulations, and formation protocols are highly co-optimized with the silicon material. As a result, introducing a new silicon supplier is closer to integrating a new cell platform than swapping in an incremental graphite source.
Furthermore, silicon processing often relies on hazardous precursors (such as silane in certain gas-phase processes) and advanced equipment. This raises commissioning and operational risk: delays in safety permitting, training, or equipment delivery can push back capacity ramps. For graphite, the equipment set—mills, reactors, furnaces—is demanding but more conventional in the materials-processing world.
4.4 ESG, Lifecycle, and Compliance Risk
ESG considerations are reshaping both graphite and silicon trajectories. Lifecycle assessments show that synthetic graphite’s energy-intensive production can have a substantial carbon footprint unless powered by low-carbon electricity. Natural graphite mining brings its own land use, biodiversity, and water-management challenges, which are increasingly scrutinized by regulators and financiers.
Silicon-anode materials are produced in relatively smaller volumes today, but their pathways often intersect with high-purity chemicals, specialty gases, and semiconductor-like operations. This can be an ESG advantage or liability depending on how energy and chemicals are sourced and managed. Some silicon-anode developers explicitly position their facilities in regions with abundant low-carbon power (e.g., hydro or renewables) to anchor a favorable lifecycle profile.
Compliance risk is evolving rapidly. Origin rules in major EV markets increasingly track not only where ore was mined, but where intermediate refining and final active-material production take place. For graphite, this can effectively restrict the share of Chinese-refined material in certain supply chains, even if technically available. For silicon, qualification of facilities in aligned jurisdictions can unlock preferential treatment, but also ties the business model to the durability of those policy frameworks.
5. Operational Realities: How Graphite and Silicon Coexist on the Line
5.1 Hybrid Anodes as the Dominant Transitional Form
The dominant near-term configuration in EV cells is not pure silicon but hybrid graphite–silicon anodes. Blending a modest proportion of silicon into graphite, often supported by modified binders and electrolytes, can deliver meaningful energy-density and sometimes power-density gains without forcing a wholesale redesign of manufacturing lines.
From a manufacturing standpoint, hybrid anodes allow existing slurry-mixing, coating, drying, and calendaring infrastructure to be retained with calibrated adjustments. Coating weights, solvent systems, and drying profiles often require optimization to handle different rheology and gas-evolution characteristics, but the capital envelope remains recognizable. This explains why many automakers are first introducing silicon in higher-end models or specific packs as a performance differentiator while keeping their main GWh volumes on conventional graphite.
5.2 Quality Control and Failure Modes
Quality control regimes differ subtly between graphite and silicon-containing anodes. For graphite, particle-size distribution, surface area, tap density, and impurity levels are the critical descriptors; variation in these dimensions affects first-cycle efficiency, rate capability, and lifetime, but the mapping from property to performance is relatively well known.

Silicon introduces additional variables: internal porosity, distribution of silicon within carbon matrices, oxide layer thickness, and the mechanical integrity of composites under cycling all need to be monitored. Standard powder metrics are necessary but not sufficient; advanced characterization—SEM/TEM imaging, in situ dilatometry, and detailed gas-evolution monitoring—play a more central role in debugging field issues.
Failure modes also shift. Graphite-dominated cells often fail through gradual SEI thickening, lithium inventory loss, or lithium plating in corner cases. Silicon-rich cells can fail via particle pulverization, contact loss, accelerated gas generation, and swelling, which can manifest as cell bloating or stack delamination. For pack engineers and safety teams, this means different surveillance and diagnostic strategies for fleets that transition from graphite-heavy to silicon-enhanced chemistries.
5.3 Capex and Process Complexity
On the anode-manufacturing line itself, the shift from graphite to silicon blends primarily affects upstream active-material supply and some aspects of slurry preparation and formation cycling. Coating, drying, and cell assembly infrastructure can often handle both, provided mechanical stability and gas management constraints are respected.
The more substantial capex implications sit at the silicon processing stage. Building a silicon-anode-material plant that handles hazardous gases, high-purity powders, and intricate thermal profiles is closer to a specialty-chemicals or semiconductor plant project than to a conventional mineral-beneficiation plant. Longer lead times for critical equipment, more complex permitting, and specialized workforce requirements all lengthen the path from project announcement to stable output.
This contrast leads to a structural dynamic: graphite’s capex intensity is skewed toward tonnage and purification; silicon’s capex intensity is skewed toward precision and safety. Both have cost implications, but the risk profile of delays and ramp curves looks different for each.
6. Scenario Lenses for 2025–2030: How the Risk Balance Could Shift
6.1 Slow Silicon, Sticky Graphite
One plausible scenario is that silicon-anode technologies progress more slowly than promotional timelines suggest. Under this path, graphite remains dominant well into the next decade, with silicon confined to premium segments or specialty applications. Diversification away from Chinese refining occurs, but at a measured pace shaped by permitting, ESG requirements, and cost competitiveness of new facilities.
In such a scenario, supply-chain resilience efforts focus on building non-Chinese spherical graphite capacity, qualifying multiple natural and synthetic sources, and optimizing synthetic-graphite energy footprints. Silicon retains a strategic role as a future option and as an energy-density booster in select platforms, but graphite’s maturity continues to anchor bulk EV production.
6.2 Hybrid Era: Silicon Blends as the New Normal
A second, increasingly likely scenario is a “hybrid era” in which silicon-graphite blends become standard in mid- to high-end EVs and gradually permeate into mass-market platforms. In this world, anode lines routinely handle formulations with modest silicon content, and qualification frameworks evolve to treat silicon vendors more like conventional material suppliers than experimental partners.
Here, graphite tonnage demand remains substantial—because even a silicon-enhanced anode typically retains a graphite backbone—but the sensitivity to any single country’s refining policies declines as silicon volumes ramp in parallel. Risk managers focus less on a binary graphite-versus-silicon choice and more on balancing portfolios of graphite sources, silicon technologies, and regional processing footprints to satisfy both performance and origin requirements.
6.3 Policy-Pulled Silicon: Industrial Strategy as a Catalyst
A third scenario sees industrial policy playing a catalytic role in accelerating silicon adoption. Subsidies, local-content rules, and defense or strategic-program demand could pull silicon-based chemistries into earlier and broader deployment than a purely techno-economic analysis would justify at this stage of maturity.
This path reshapes the risk balance: silicon’s technology and scaling risk becomes more prominent in the near term, but geopolitical and compliance risks tied to graphite decline more quickly. The system effectively exchanges one category of risk (concentration and policy exposure) for another (technology performance and ramp reliability). Whether this exchange is favorable depends on each manufacturer’s product mix, regional exposure, and risk tolerance.
Across all scenarios, one meta-conclusion stands out: energy-density gains from silicon do not automatically neutralize graphite risk; they re-weight it. Even aggressive silicon adoption leaves graphite as a significant component of global anode mass for years, meaning that diversification and ESG-driven reforms in graphite supply chains remain strategically relevant alongside silicon’s rise.
Conclusion: Trade-Offs, Not Silver Bullets
Graphite versus silicon in anodes is often framed as a replacement story. The operational reality is more nuanced. Graphite underpins the current lithium-ion system with known behavior and extensive industrial infrastructure but is exposed to refining concentration and ESG scrutiny. Silicon offers step-change performance potential and a more diversified, policy-aligned geographic base, but introduces unresolved questions around scaling, long-term durability, and complex processing.
For the remainder of the decade, the anode landscape is likely to be defined by co-existence: graphite-heavy chemistries, graphite–silicon hybrids, and niche high-silicon formats operating side by side. The strategic differentiator will not be a single chosen chemistry, but the ability to orchestrate material portfolios, regional processing, and qualification roadmaps in a way that keeps performance, cost, and compliance in balance.
Materials Dispatch will continue to track weak signals across three fronts: refinements in silicon-anode architectures and their field data, the build-out and policy treatment of graphite refining outside China, and regulatory shifts that redefine which materials “count” toward critical-mineral benchmarks. The intersection of these signals will determine how the graphite–silicon risk map evolves from technical promise to industrial reality.
Note on Materials Dispatch methodology Materials Dispatch integrates continuous monitoring of regulatory communications (including agencies such as MOFCOM and Western trade authorities), company disclosures on graphite and silicon projects, and end-use performance specifications from EV and storage platforms. This triangulation enables a technology-grounded read of where material science, industrial capacity, and policy constraints genuinely intersect in the graphite–silicon anode transition.
Anna K
Analyste et rédacteur chez Materials Dispatch, spécialisé dans les matériaux stratégiques et les marchés des ressources naturelles.


