Tech Deep Dive: Processing Flowsheets for Gallium, Germanium, and Rare Earths
Processing flowsheets for gallium (Ga), germanium (Ge), and rare earth elements (REEs) have moved from background engineering topics to front-line supply security issues. The core technical reality is simple: ores and byproducts are increasingly accessible, while proven, compliant, and scalable processing routes remain the hard constraint. The current U.S.-led projects between 2022 and 2025 illustrate this tension with unusual clarity.
The United States is rebuilding capabilities in materials where China currently dominates refined supply: refined Ga and Ge are heavily concentrated, and REEs are still largely processed through Chinese-controlled solvent extraction (SX) hubs. DOE-funded programs, university-industry pilots, and national lab initiatives are testing a new generation of flowsheets using coal byproducts, acid mine drainage (AMD), and unconventional carbonatites. This article dissects those flowsheets from an operational perspective: unit operations, energy and reagent demands, impurity management, and the failure modes that emerge when bench chemistry hits continuous pilot scale.
Across Ga, Ge, and REEs, the pattern is consistent. Leaching and initial dissolution are relatively mature. Real bottlenecks appear where selectivity, phase behavior, and equipment reliability intersect: co-precipitation in pH-controlled impurity removal, SX organic degradation under real impurity loads, and electrode or membrane fouling in advanced electrochemical systems. These are not academic issues; they directly determine whether domestic projects can meaningfully offset China-centric processing in the medium term.
1. Context and the Operational Question
Export licensing controls on Chinese gallium and germanium products from 2023 onward exposed how concentrated these supply chains had become. Public data and industry statistics show that China accounts for the overwhelming majority of refined Ga and Ge output and a dominant share of REE separation capacity. At the same time, U.S. and allied industrial policy-through instruments such as the Bipartisan Infrastructure Law (BIL) and targeted DOE funding calls-has pushed for domestic or friendly-jurisdiction recovery from secondary feeds: coal/lignite, zinc residues, AMD, and carbonatite deposits.
The operational question is no longer whether gallium, germanium, and REE units exist in those feedstocks. They clearly do, typically in the tens to hundreds of ppm range for Ga and Ge and percent-level for REE oxides in enriched ores and concentrates. The question is whether flowsheets can deliver consistent, on-spec material at industrially relevant scale without prohibitive energy, reagent, or compliance penalties. That requires a sober look at each unit operation across three intertwined systems:
- Gallium flowsheets, largely as a byproduct from zinc, bauxite, coal, and carbonatites.
- Germanium flowsheets, from zinc slags and coal-derived concentrates.
- REE flowsheets, from monazite, bastnäsite, coal byproducts, AMD, and carbonatites, often co-recovering Ga and Ge.
The following sections analyze representative flowsheets being tested in U.S. pilots and lab-pilot bridges, with specific attention to the points where laboratory yields collapse or OPEX escalates once hydrodynamics, erosion/corrosion, and real-world feed variability are introduced.
2. Gallium Processing Flowsheets: Unit Operations and Constraints
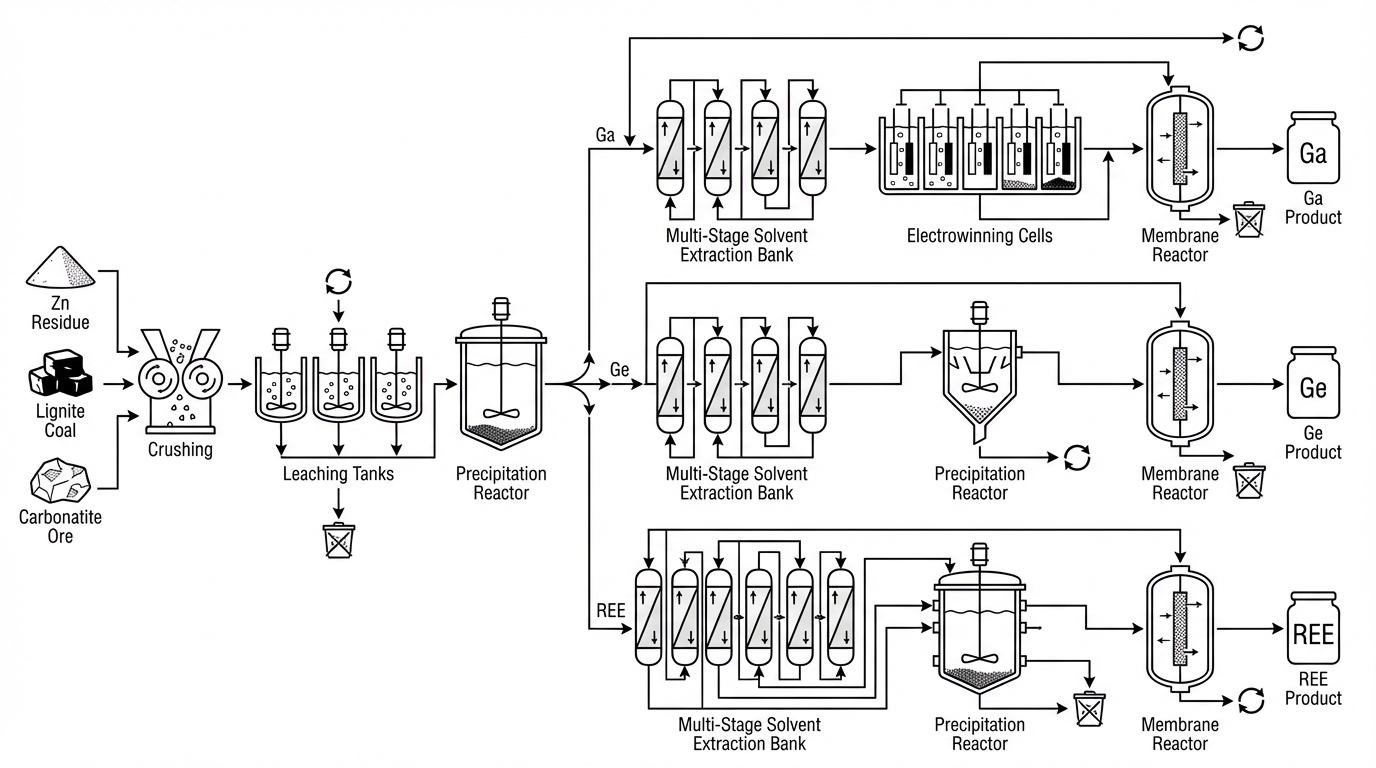
Gallium is classically a byproduct metal. It is present in bauxite (via the Bayer process), zinc refinery residues from sphalerite ores, coal and lignite ash, and certain carbonatites. Contemporary U.S. pilots draw heavily on zinc residue and coal-based flowsheets, adapted from historical European and Australian practice but updated with modern SX chemistry and emission standards. A typical zinc-residue-based flowsheet proceeds through leaching, impurity precipitation, solvent extraction, and electrowinning or cementation followed by refining.
2.1 Feedstock Preparation and Acid Leaching
Zinc refinery residues or similar intermediates, often containing Ga and Ge in the 0.1–0.5% range, are ground and conditioned for leaching. A benchmark flowsheet employs sulfuric acid leaching at elevated temperature, for example around 80 °C with moderate acid strength and an elevated liquid–solid ratio. Under optimized conditions reported in the technical literature, leaching recoveries can approach nearly complete dissolution for gallium and high but somewhat lower recoveries for germanium.
The first major constraint appears immediately: co-dissolution of iron and aluminum, often contributing more than 20% of the liquor mass. Ferric iron in particular forms strong complexes, drives viscosity up, and interferes with later SX selectivity and phase disengagement. One commonly reported mitigation is partial reduction of ferric to ferrous iron using SO₂ or similar reductants, coupled with staged neutralization. This improves downstream yields but introduces its own CAPEX and permitting footprint, including gas handling and off-gas treatment infrastructure.
From an execution standpoint, leaching is not limited by chemistry but by impurity management strategy. Plants that under-invest in front‑end impurity control find that they have simply moved the problem into larger, more complex, and more sensitive downstream circuits.
2.2 Impurity Precipitation and pH Windows
Following leaching, the liquor carries Ga and Ge alongside a broad suite of base metal impurities (Al, Fe, Zn, Cd, Pb, Cu, and others). Standard practice uses staged pH elevation with calcium hydroxide, magnesium compounds, and sodium hydroxide to precipitate these impurities as hydroxides or basic salts. In some zinc-derived flowsheets, Mg-based reagents are used to pull germanium into a germanate-rich precipitate while leaving most gallium in solution.
The operational difficulty lies in the narrow pH windows. Published case studies show that once pH drifts above the mid‑4 range, gallium starts to co-precipitate significantly with germanium and other hydroxides, leading to losses on the order of tens of percent from the soluble Ga pool. Conversely, running too acidic allows troublesome levels of iron, aluminum, and heavy metals to pass forward. Historic pilot work (including operations at Pasminco’s former zinc complexes) documented batch rejections of a substantial fraction of production due to variable residue composition and imperfect pH control.
Automated pH control, rapid mixing, and sufficient residence time are not glamorous topics, yet they consistently differentiate stable flowsheets from those that oscillate between impurity breakthrough and co-precipitation losses. The most advanced chemistries cannot rescue a flowsheet where this basic control loop is fragile.
2.3 Solvent Extraction for Gallium Recovery
Once a reasonably clean gallium-bearing solution is prepared, SX steps in as the workhorse separation tool. Organophosphorus extractants such as D2EHPA or related phosphoric/phosphonic acids are typically deployed in multiple stages, with gallium loaded into the organic phase and then stripped with an acidic solution to yield a concentrated gallium liquor.
Bench data commonly report gallium extraction efficiencies above the mid‑90% range with similar performance in stripping. However, those figures assume reasonably benign impurity profiles. Real residues from coal-derived feeds and complex zinc sludges routinely carry arsenic, antimony, and organic matter that can degrade the organic phases, shorten SX cycle life, and promote stable emulsions. Germanium can also co-extract at the 5–10% level, forcing either a dedicated Ge SX front‑end or complicated bleed and recycle strategies. Each added SX circuit roughly doubles the organic inventory and increases sensitivity to foaming, crud formation, and solvent losses.
This is one of the recurring structural findings in current U.S. flowsheets: SX is extremely powerful, but every additional separation target and every poorly controlled impurity multiplies not just complexity but also the number of failure modes to monitor.
2.4 Electrowinning and High-Purity Gallium Refining
After SX, gallium-rich strip liquors are treated via electrowinning or cementation to recover metallic gallium. Electrowinning onto aluminum cathodes at moderate temperatures and voltages is standard. Reported current efficiencies often fall below ideal values due to hydrogen evolution, side reactions, and electrode fouling, resulting in meaningful energy consumption per kilogram of gallium produced.
Scaling from laboratory cells to multi‑tonne per year pilots tends to reveal hidden maintenance burdens. Thin gallium deposits can spall; impurities like arsenic or silica embed in cathode films; and anode sludges accumulate faster than expected. Some pilots have reported double‑digit percentages of downtime tied to cleaning cycles and electrode replacement. Final purification to 4N (99.99%) and beyond often relies on zone refining or vacuum distillation, which add additional electricity and equipment overhead but are relatively straightforward once bulk metal is in hand.
For defense‑grade GaAs semiconductor applications, these last refining steps are non‑negotiable. The upstream flowsheet is therefore judged not only on total recovery but also on its ability to deliver a feed that can be upgraded to electronics-grade without heroic batch rework.
2.5 Coal Byproduct versus Zinc Residues: Logistics and Scale
DOE-sponsored pilots such as the Microbeam–University of North Dakota program are pioneering gallium recovery from lignite and coal combustion byproducts. These flowsheets broadly parallel the zinc-residue route-acid leaching, impurity precipitation, gallium/germanium SX—but begin from very large tonnages of low-grade material. While this path avoids the long timelines and permitting complexity of new mines, it introduces a logistics problem: thousands of tonnes of lignite or ash need to be moved, stockpiled, and fed to central plants for relatively small amounts of Ga and Ge.
Rail and trucking requirements, material handling infrastructure, and weather-related disruptions become non-trivial OPEX drivers. Internal analyses from project partners indicate that logistics alone can add double‑digit percentage uplifts to operating costs compared with treating concentrated zinc residues. Yet this approach can capitalize on existing power-sector waste streams and offer a compelling route to small but strategically important quantities of gallium concentrates in the low- to mid‑single‑digit MT/year range.
3. Germanium Processing Flowsheets: Front-Loaded Selectivity
Germanium flowsheets resemble gallium flowsheets but are typically structured to recover Ge earlier and more aggressively, reflecting its role in fiber optics, infrared optics, and specialty electronics. Ge-bearing zinc slags, coal/lignite ash, and certain polymetallic concentrates are the primary feeds in current U.S. programs, including the same Microbeam–UND integration where Ge and Ga are co-recovered from lignite-based REE concentrates.
3.1 Leaching of Ge-Bearing Feeds
Acid leaching with sulfuric or hydrochloric acid at elevated temperatures is again the starting point. Two-stage leaching sequences are often reported as optimal: a first aggressive leach to dissolve easily accessible germanium, followed by a second, somewhat milder stage to avoid excessive gangue dissolution. Recovery ranges into the 80–90% bracket have been cited for optimized circuits.
However, feeds containing significant siderite (FeCO₃), common in some coal measures, buffer the acid and depress effective leaching efficiency by notable margins. Pilot work has shown that pre-roasting at temperatures around 600 °C can decompose carbonate phases, improving subsequent leach performance but generating CO₂ and SO₂ streams that complicate air permitting under tightened emissions frameworks. Roaster CAPEX and fuel costs add further friction.
3.2 Precipitation and Early Germanium Capture
Germanium is often captured as a hydrated oxide or germanic acid intermediate. Classical routes employ tannic acid, magnesium salts, or other organic–inorganic reagent systems to selectively precipitate germanium around pH values in the 4–5 range. Reported yields for these steps are high when the solution chemistry is well controlled.
The tradeoff is that gallium and residual base metals can co-precipitate. Historic operations documented gallium co-precipitation in the 10–15% range when Ge was pulled early without prior SX separation. This may be acceptable where gallium is a minor byproduct, but becomes problematic in integrated Ga–Ge flowsheets. As with gallium, narrow pH windows and tight control of reagent dosing determine whether these precipitation steps deliver selective capture or simply generate mixed hydroxide sludge that requires re-treatment.
3.3 Chlorination, Distillation, and Metal Production
Once germanium is present as oxide, conversion to germanium tetrachloride (GeCl₄) followed by distillation remains a cornerstone of high-purity Ge production. Chlorination at elevated temperatures generates volatile GeCl₄, which can be distilled at relatively low boiling points and then hydrolyzed back to high-purity GeO₂. Subsequent reduction with hydrogen or magnesium yields metal.
The technical and operational challenges center on corrosion and gas handling. Chlorine-containing environments at high temperature demand specialty alloys such as Hastelloy for reactors and piping, significantly lifting CAPEX compared with purely hydrometallurgical routes. In addition, some U.S. pilot work has reported non-trivial vapor losses of GeCl₄ during scale-up, pushing overall yields down from laboratory expectations. Gas capture, scrubbing, and condensate management systems therefore become central to flowsheet robustness rather than peripheral add-ons.
Another critical constraint is hydrogen purity during final reduction. Sub‑ppm levels of oxygen, moisture, or hydrocarbons can introduce defects in optical-grade GeO₂ used for fiber optics. This pulls in high-spec gas purification, leak detection, and quality assurance infrastructure that sits well beyond conventional base-metal metallurgy.
3.4 Variability and Real-Time Characterization
Germanium concentrations in coal and lignite feeds can vary substantially, with ranges of tens to hundreds of ppm within a single mine or seam. The DOE-funded Microbeam–UND project explicitly addresses this issue by combining beneficiation, XRF/LIBS sensing, and dynamic blending to stabilize feed quality into the hydromet circuit. Commissioning timelines reported for such integrated setups underline an important lesson: analytical infrastructure and control logic can be as gating as any reactor or SX mixer-settler.
In essence, germanium flowsheets are a stress test of a project’s analytical discipline. Where feed characterization and process control are strong, Ge recovery steps can run close to bench expectations. Where they are weak, variability cascades into under- or over-dosing of reagents, phase instability, and ultimately wide swings in product purity.
4. Rare Earth Elements: Complex SX Cascades and New Electrochemical Routes
Rare earth element processing sits at the heart of contemporary critical materials debates. While Ga and Ge flowsheets involve recovering ppm-level byproducts from base-metal or coal circuits, REE flowsheets deal with percent‑level REO concentrates but face the opposite challenge: separating 17 chemically similar elements to multiple purity tiers across different end uses. U.S. projects under BIL and DOE FOAs are revisiting both classic solvent extraction approaches and newer electrochemical and membrane routes.

4.1 Beneficiation and Leaching of REE Ores and Byproducts
Classical hard-rock deposits such as bastnäsite and monazite undergo crushing, grinding, and flotation to yield REO concentrates. Carbonatite deposits like Sheep Creek in Montana, pursued by US Critical Materials, have reported total rare earth grades in the high‑teens percent range and notable gallium credits in the 180–385 ppm band. Coal-based REE projects instead target fly ash, bottom ash, or specially processed coal-derived concentrates, typically with REE grades in the thousands of ppm. AMD projects treat large throughputs of acidic drainage with dissolved REEs at lower concentrations but continuous flow.
Leaching chemistries vary with mineralogy. Sulfuric acid under elevated temperature and sometimes pressure is standard for many carbonatites and monazites, while hydrochloric systems may be favored for coal and AMD streams to facilitate chloride-compatible downstream separation. Fluoride-bearing feeds such as bastnäsite introduce another layer of complexity: formation of HF can drive severe corrosion, requiring titanium-lined autoclaves and upgraded ventilation and scrubbing, adding materially to plant CAPEX.
4.2 Group Separation via Solvent Extraction
Once in solution, REEs are commonly separated into light (LREE) and heavy (HREE + Y) groups via SX with extractants such as HEH(EHP) (often cited as P507). Light rare earths load more readily, enabling initial separation into a light-enriched organic phase and a heavy-enriched raffinate. Multiple mixer-settler stages (often in the range of several per group step) and complex pH gradients are used to sharpen separation.
The selectivity factors between adjacent rare earths in these systems, however, are modest—often low single-digit numbers. Achieving high-purity oxides (99.9% and above) for dysprosium, terbium, neodymium, and others requires cascades of hundreds of stages when using conventional solvent and configuration choices. Energy usage per unit volume of liquor handled and organic solvent losses become structurally significant. Emulsion formation, crud accumulation, and phase inversion events are recurrent operational risks, particularly for coal-derived and AMD feeds carrying organic matter, sulfate soaps, and fine solids.
This is where the gap often appears between planning documents and reality: theoretically elegant SX trains need to confront the fact that every additional stage is another chance for mechanical or chemical instability to propagate.
4.3 Individual Separation, Precipitation, and Calcination
After group splits, individual rare earths are produced through finer-grained SX circuits, ion exchange, or combinations thereof. Oxalic acid or carbonate precipitation then converts REE-bearing solutions into solid intermediates, which are calcined at temperatures around 800 °C to yield REE oxides. Further metal production uses metallothermic reduction or, in advanced research programs, ionic liquid electrolysis and plasma-based processes.
Each additional separation step carries a tradeoff between purity, recovery, and plant complexity. For example, mid‑REEs such as samarium and gadolinium can exhibit poorer stripping efficiency than the lightest or heaviest lanthanides, driving up recycle flows and solvent inventory. Field data suggest double‑digit annual solvent losses in some pilot operations, underlining the importance of solvent regeneration and waste handling strategies both for OPEX and for environmental compliance.
4.4 Electrochemical Membrane Reactors: Promise and Constraints
An emerging variant is the electrochemical membrane reactor (EMR) approach being developed by Idaho National Laboratory with US Critical Materials for carbonatite leachates containing both REEs and gallium. In this concept, electrical potential, water, and nitrogen are used to drive selective transport and recovery of target metals without large volumes of organic solvents or classical extractants. Project communications indicate targeted REE and Ga recoveries exceeding 90% at bench scale.
Early data show notable practical constraints. Silicate and other colloidal impurities in carbonatite leachates tend to foul membranes, reducing effective membrane life to hundreds of hours before cleaning or replacement is required. Overpotentials at industrially relevant current densities increase energy consumption per unit of REE recovered compared with optimized SX. Gas handling, electrode materials, and scaling behavior across large membrane areas are unresolved at industrial scale.
The key insight is that EMR-style systems trade solvent management and large SX hall footprints for membrane integrity and electrochemical stability challenges. They do not eliminate complexity; they rearrange it.
4.5 Co-Recovery of Other Critical Minerals
Several AMD and coal-based REE projects aim to co-recover lithium, cobalt, nickel, gallium, and germanium. While technically attractive, this multi-target strategy can strain selectivity. For example, extractants tuned for lithium can exhibit significant loading of certain rare earths, contaminating lithium product streams and complicating downstream carbonate or hydroxide production. Conversely, REE-centric SX circuits may drag lithium or transition metals into raffinate or intermediate phases where they are harder to recover efficiently.
The lesson from the most advanced flowsheets is that parallel circuits and selective bleed streams, rather than simple “catch-all” extractant systems, tend to offer more controllable outcomes—even at the expense of higher apparent complexity. Attempts to solve too many separation problems in a single SX or IX circuit often build in chronic cross-contamination that is expensive to remove later.
5. Integrated Ga–Ge–REE Flowsheets in Current U.S. Pilots
Several U.S. initiatives now integrate gallium, germanium, and REEs within single flowsheets, aiming to extract maximum value from unconventional feeds.
Microbeam–UND lignite project (North Dakota). This DOE-funded effort processes lignite-derived REE concentrates via acid leaching, followed by SX circuits for Ge and Ga, and then REE separation. Conceptual designs target concentrated Ge/Ga outputs in the single-digit MT/year range from a feed of roughly hundred‑tonne‑scale MREE concentrates per year. Technical disclosures highlight feed variability in Ge and Ga content (tens to hundreds of ppm), driving the adoption of real-time LIBS/XRF sorting and blending before leaching. Process flow diagram finalization has reportedly lagged behind initial timelines due to the need to stabilize this front‑end variability.
US Critical Materials–INL Sheep Creek carbonatite program (Montana). Here, high-grade carbonatite ore with substantial REE and gallium content is treated via EMR-based electrochemical recovery instead of classical SX. Publicly available materials present a “no additional reagents” vision using electricity, water, and nitrogen. Practically, this still entails careful control of gas purity (for nitrogen and other process gases), electrode materials, and pre-filtration to limit membrane fouling from silicate and carbonate particulates. External industrial gas supply logistics, especially in remote locations, become as critical as ore hauling.
AMD-based REE and co-product recovery under DOE FOA 2619. Selected projects process sizeable AMD flows—hundreds of gallons per minute—through SX-based circuits designed to produce tens of tonnes per year of REE products, with side-streams aimed at Ga and Ge recovery where feed chemistry allows. These flowsheets bypass greenfield mining and instead turn a legacy environmental liability into a critical-mineral source. At the same time, permitting for AMD capture and treatment infrastructure often front-loads two or more years of engagement with environmental regulators, landowners, and existing mine operators.
Across all three models, two cross-cutting constraints dominate: impurity management (As, Sb, silica, organic matter) that degrades SX and membranes, and the systematic loss of recovery when scaling from beaker to continuous pilot. Bench-top yields in excess of 90% often translate to 70–80% in pilot operation once real hydrodynamics, phase disengagement times, and recycle loops come into play.
6. Common Constraints and Operational Tradeoffs
Despite differences in feedstocks and target products, gallium, germanium, and REE flowsheets encounter a common set of technical choke points. These can be summarized by unit operation, typical constraint, and operational impact.
| Unit Operation | Typical Constraint | Impact on Yield / OPEX | Observed Mitigation Approaches (2022–2025 U.S. Pilots) |
|---|---|---|---|
| Leaching | Co-dissolution of Fe/Al and carbonate buffering | Yield loss and higher downstream reagent demand | Pre-roasting, reductive conditions, staged acid addition |
| pH-Controlled Precipitation | Narrow pH windows; co-precipitation of Ga/Ge | Batch rejections; Ga/Ge losses in sludges | Multistage pH cascades, improved mixing, real-time pH and ORP monitoring |
| Solvent Extraction (Ga/Ge/REE) | Limited selectivity between similar species; emulsions; organic degradation | More stages; higher energy and solvent makeup; downtime | Optimized extractant systems, demulsifiers, continuous crud management |
| Electrowinning / Electrolysis | Electrode fouling; side reactions (H₂ evolution) | Lower current efficiency; maintenance-driven downtime | Pulse current regimes, refined electrolyte chemistry, scheduled cleaning cycles |
| Electrochemical Membrane Reactors | Membrane fouling by silicates and organics; overpotentials | Shorter membrane lifetimes; higher kWh per kg metal | Pre-filtration, slurry conditioning, membrane module redundancy |
A recurring pattern emerges from this comparison. Leaching is typically not the rate‑limiting step; relatively standard chemical engineering approaches can achieve high dissolution rates. Instead, the constraints are dynamic: pH drift, phase behavior in SX, and physical fouling in electrochemical units. In other words, what appears as a purely “chemical” problem on paper is often a control and materials-handling problem in the real plant.
There is also a clear tradeoff between reagent intensity and equipment intensity. Classic hydrometallurgical flowsheets (acid leaching + SX + precipitation) consume significant reagents and generate sizable liquid and solid wastes but rely on well-understood, relatively forgiving equipment. Newer EMR or membrane-based approaches aim to reduce reagents and waste volume at the cost of high-spec membranes, more sophisticated electrochemical control, and tighter water quality requirements.
7. Compliance, Environmental, and Logistical Realities
Environmental compliance is not a parallel track; it directly shapes flowsheet design. Rare earth and Ga/Ge circuits inherently involve acids, bases, chloride or sulfate systems, and potentially toxic impurities like arsenic and cadmium. Wastewater treatment, neutralization, solid residue stabilization, and air emissions control define not only permitting timelines but also long-term operating risks.
In the United States, major DOE-supported pilots fall under NEPA review, with environmental assessments and, in some cases, full environmental impact statements. These can stretch timelines by several quarters but also create a framework for robust water management, tailings or residue handling, and emissions control. Projects introducing novel reagents or extractants may also intersect with TSCA requirements, particularly if organic SX systems or ionic liquids fall outside existing regulatory experience.
On the logistical side, the contrast between centralized, high-grade operations and distributed, low-grade feed utilization is stark. AMD and coal byproducts offer short lead times and no exploration risk, but they imply high volumes, dispersed sites, and sensitive stakeholder relationships (utilities, mining legacies, landowners). Carbonatite or hard-rock REE operations carry more traditional mining footprints but can deliver far higher grades and simpler logistics for the processing plant, at the cost of longer mine permitting and development sequences.
Resilience-oriented analysis therefore often focuses less on headline capacities and more on the vulnerability of each flowsheet to single-point failures: a rail line outage for lignite shipments, a nitrogen supply disruption for EMR-based gallium circuits, or a SX organic supplier issue for REE separation plants.
8. Scenarios and Structural Options for Ga, Ge, and REE Processing
Considering the technical and regulatory landscape, several structural configurations for Ga, Ge, and REE processing are emerging in North America and allied jurisdictions:
- Byproduct-centric hubs. Zinc, aluminum, and coal-related sites add Ga/Ge/REE recovery circuits, leveraging existing infrastructure and permitting but dealing with complex impurity suites and logistics.
- Dedicated REE + Ga carbonatite plants. High-grade deposits such as Sheep Creek anchor integrated plants that use SX, EMR, or hybrids to co-produce REE oxides and gallium concentrates, with germanium potential where present.
- AMD treatment clusters. Regional AMD sources feed centralized processing, allowing modular expansion and flexible feed blending at the expense of strong dependency on environmental and regulatory frameworks.
Historically, similar patterns were seen in the evolution of niobium and tungsten processing. Early niobium production tied to pyrochlore projects was dominated by a handful of integrated mines with captive processing, while secondary recovery from slags and byproducts struggled to find stable flowsheets. Tungsten has likewise oscillated between mine-centered and scrap/byproduct-centered supply, with flowsheet complexity often determining which route dominated at any given time.
The key structural insight is that flowsheets act as amplifiers of upstream volatility. Flexible, impurity-tolerant flowsheets can accommodate variable byproduct streams and incremental expansions. Highly optimized but narrow-window flowsheets deliver excellent economics under ideal conditions but are brittle under feed or regulatory shocks. Ga, Ge, and REE projects now being built will reveal over the next few years where along this spectrum the current generation of technologies truly sits.
9. Conclusion: What Really Governs Ga, Ge, and REE Flowsheet Performance
Across gallium, germanium, and rare earth processing, one pattern recurs: geology sets the stage, but hydrometallurgical and electrochemical flowsheets decide industrial reality. High leach recoveries on paper do not guarantee viable supply; the decisive factors are impurity management, SX and membrane stability, and the interaction between control systems and variable feeds.
Hydrometallurgical circuits with extensive SX deliver high purity and proven scalability but carry heavy reagent, water, and waste burdens. Electrochemical and membrane-based innovations promise leaner reagent footprints and potentially smaller environmental stacks but transfer complexity into materials science and electrochemical control. Integrated Ga–Ge–REE flowsheets increase value density yet multiply interfaces where instability can emerge.
Materials Dispatch tracks these developments as indicators of future supply resilience, watching not only headline announcements but also weak signals from pilot data, permitting documentation, and technical disclosures from programs such as DOE FOA 2619 and related initiatives. The way these early flowsheets handle impurities, scale-up losses, and regulatory constraints will quietly determine how much of the Ga, Ge, and REE supply chain truly diversifies in the coming decade.
Note on Materials Dispatch methodology Materials Dispatch integrates patent filings, technical papers, DOE and USGS reporting, and policy releases from entities such as MOFCOM to reconstruct how flowsheets evolve in practice, not just in design. This article combines open technical data on Ga/Ge/REE processing with analysis of end-use purity requirements in semiconductors, magnets, and optics to identify where operational bottlenecks are likely to emerge first.
10. Sources and Further Reading
- NETL / DOE Project FE0032124 – Microbeam Technologies and University of North Dakota lignite-based REE, Ga, and Ge recovery project documentation.
- Technical papers on gallium and germanium recovery from zinc refinery residues and coal byproducts presented at international Pb-Zn and ICSOBA conferences.
- US Critical Materials and Idaho National Laboratory materials on electrochemical recovery of gallium and rare earths from Sheep Creek carbonatite.
- DOE BIL Critical Minerals FOA 2619 project selection summaries for REE and critical mineral advanced processing.
- USGS Germanium statistics and related critical mineral assessments detailing global production and refining concentration.
- Recent solvent extraction optimization studies for coal-based REE streams in peer-reviewed chemical engineering journals.
Anna K
Analyst and writer at Materials Dispatch, specializing in strategic materials and natural resources markets.



